Articles
Volumetric or Coulometric: Which Karl Fischer titration method is better?
posted : 15 آذر 1404
To determine the amount of water in a sample and obtain accurate results, choosing the optimal Karl Fischer titration method between volumetric and coulometric is essential. The purpose of this brief text is to assist those who need to choose between the two methods while have not much experience working with Karl Fischer devices.
Introduction:
Karl Fischer titrators determine water content using two different methods: volumetric and coulometric. Obviously, selecting the appropriate method between the two requires understanding the advantages and limitations of each.
In both methods, iodine is used as the reagent reacting with water, but the fundamental difference between them lies in the mechanism and how iodine is added to the titration vessel to react with water.
In the volumetric method, iodine is added to the in the titration vessel and reacts with the sample’s water content. The titration endpoint is reached when the reaction of iodine with water stops due to the complete consumption of the sample’s water content. In this method, the amount of water in the sample is calculated based on the volume of Karl Fischer reagent consumed. The accuracy and acceptable precision of the volumetric Karl Fischer method depend on the ability to perform titrations with very precise and repeatable dosages. Then, regularly checking the Karl Fischer solution to ensure its freshness and adequacy is essential when using this method.
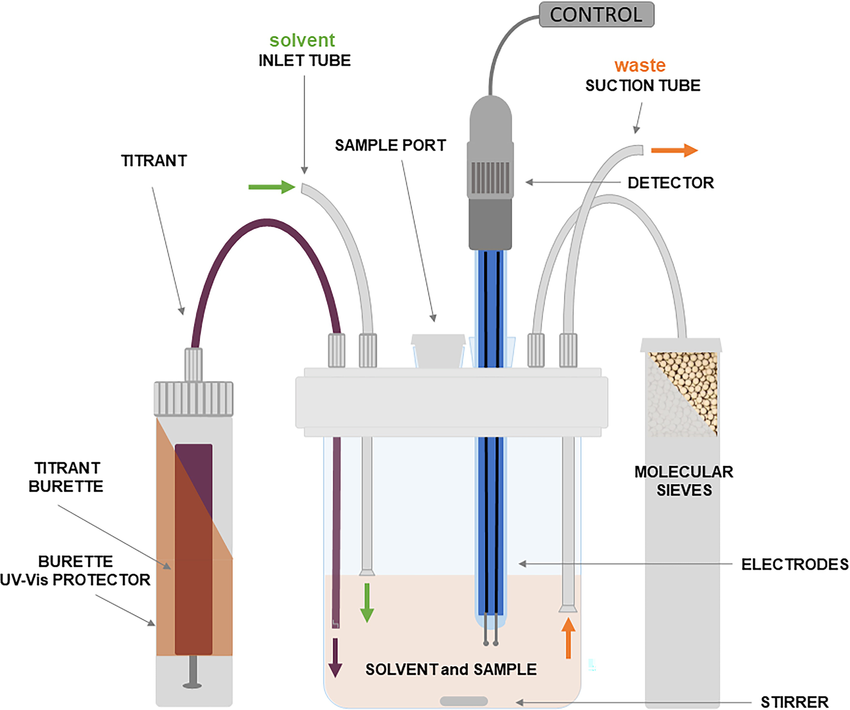
Fig.1. Volumetric KF Vessel
In the coulometric method, instead of using Karl Fischer solution and performing precise doses, an electric current is used to releases the iodine from the iodine-containing reagent needed to react with the water present in the sample, so that each water molecule consumes one released iodine molecule. As soon as the water is completely consumed a small amount of un reacted iodine molecules remain in the solution, causing a significant decrease in the solution's resistance which sets the end point of the titration. In this method, the amount of water in the sample is calculated by multiplying the titration time by the current required to reach the endpoint.
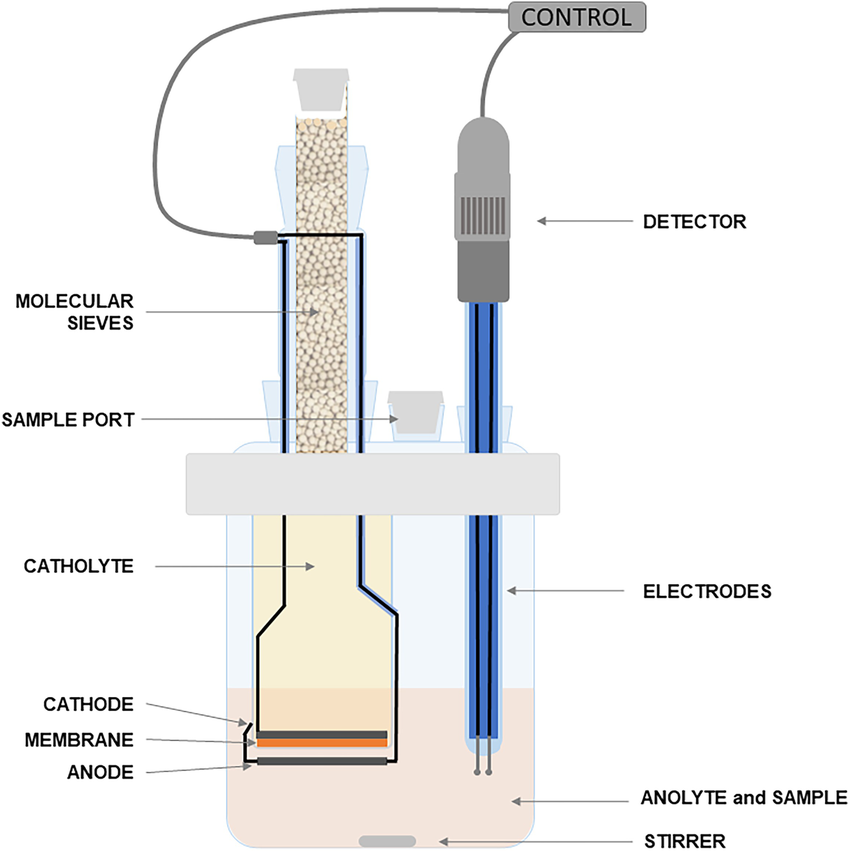
Fig 2. Coulometric KF Vessel
Now let us come back to the question: Which method is more suitable for determination of water content; Volumetric or Coulometric Karl Fischer titration?
The choice between the two Karl Fischer methods, depends on various factors as follows:
A) The amount of water content
The volumetric method is used for the water content ranging from 0.01 to 100 percent (100 ppm and above), while Karl Fischer coulometric method is used for the water content between 0.001 to 0.1 percent ( 10 ppm to 100 ppm). Of course, these numbers are not absolute. In other words, it might be possible to use either method for determining water content in a range different from what is mentioned, but the optimal range for their application is:
Volumetric KF: 0.01 to 100 percent
Coulometric KF: 0.001 to 0.1 percent
B) Physical form of the sample
In the volumetric method, it is possible to open a valve on the lid of the titration vessel, but this is not possible in the coulometric method. Considering this, if the sample is in liquid form, water content determination can be done using either method because the liquid sample can be injected with a syringe through a septum without opening the lid of the titration vessel. However, a solid sample can only be added by opening the lid of the vessel; therefore, if the sample is solid and powdery, the volumetric method is preferred.
C) Sample Dissolution Capability
Accurate and precise determination of water content in a sample by the Karl Fischer method requires its complete dissolution in the solvent and full reaction with the Karl Fischer reagent. If the sample does not dissolve well in the solvent, some methods must be used to aid its dissolution. One of these methods is increasing the temperature, which can be achieved by changing the titration vessel from a regular type to a double-walled type with a thermostatic cover. This can be done in both volumetric and coulometric KF methods.
Another method to aid dissolution is applying a homogenizer in the titration vessel. Using a homogenizer is possible during titration in the volumetric method, while in the coulometric it can only be done before titration and in a separate container.